
Symbiosis has a proven track record of supporting GMP aseptic fill finish for recombinant proteins and monoclonal antibody manufacturing.
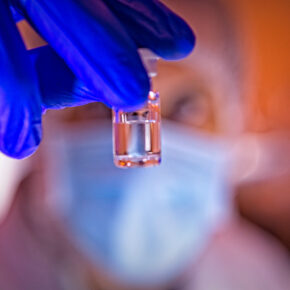
Over the past 40 years recombinant proteins have risen to become a core part of the therapeutic landscape. From relatively small hormones, to large and complex monoclonal antibodies (mAbs), the manufacturing process is now well characterised and well understood.
However, consideration must still be given to how the therapy will be delivered to the patient and therefore what the final product format will be.

Symbiosis has extensive experience in delivering many hundreds of clinical projects related to mAbs and recombinant proteins. We can support your clinical trials.
Symbiosis are the leading fast access CMO for clinical or small scale commercial manufacture and our offering is built upon the following principles:
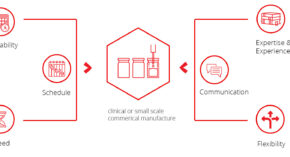
Waiting 6 or even 12 months for a filling slot can be detrimental. We work to your schedule and strive to have availability in as little as 8 weeks
Delivering your product to the clinic on time is essential, given patients are often waiting. Therefore, we execute over 90% of projects within the agreed timeframe.
We understand the need for results is a critical factor in the progression of clinical trials. Our typical fill-finish campaign takes 12 weeks.
Our dedicated team has handled >600 filling campaigns for mAbs to more challenging ATMPs for >100 customers. You are in safe hands.
Our service is underpinned by our expert project management team who will keep you informed every step of the way.
Drug development and manufacturing occurs in a dynamic environment. We work flexibly, where possible, to accommodate any changes that you may require.